|
This is because they cancel each other’s waves due to the anti-phase. Because of the anti-phase, the waves in the middle of the hydrogen atoms are completely zero. In contrast, what about the anti-phase wave? In the case of anti-phase, the situation is different from the previous one. The Anti-Bonding Orbitals (LUMO) Have Nodes and High Energy The result is the creation of bonds between atoms. The bonding orbitals (HOMO) are created by combining orbitals that are in phase. The electrons help the hydrogen molecules to form bonds. However, since electrons have a negative charge, the presence of electrons between hydrogen atoms creates a stable state and allows them to form bonds with each other. In other words, the atoms bond with each other to form a molecule.Ītoms have protons. When two hydrogen atoms of the in-phase come close to each other, the two hydrogen atoms will overlap. A hydrogen atom has a single electron and an s-orbital. Also, it is the anti-bonding orbitals that are involved in the cleavage of the molecular bond.įirst, let’s consider the example of the hydrogen molecule. What is the difference between a bonding orbital and an anti-bonding orbital? It is literally the bonding orbitals that are involved in the bonding of the molecule. On the other hand, LUMO stands for Lowest Unoccupied Molecular Orbital, and LUMO refers to anti-bonding orbitals. HOMO stands for Highest Occupied Molecular Orbital, and HOMO refers to a bonding orbital. Once we understand that electrons have properties as waves and that they enhance or cancel each ‘s properties, we can understand the concept of HOMO and LUMO. Difference Between Bonding and Anti-bonding Orbitals Rather, we can understand that electrons are a type of wave. The term “wave function” can be difficult for us to understand. On the other hand, when waves of opposite phase are combined, they cancel each other out, and no vibration occurs. When waves of the same phase are combined, the waves become larger because they overlap with each other. Electrons are a type of wave, and we use the wave function.Īs you can see from the waves in the ocean, the waves ripple up and down. In quantum mechanics, it is represented by a wave function. Chemical reactions involve HOMO and LUMO.In simple terms, the following is the frontier startup theory. In quantum mechanics, there is something called a frontier orbital theory. Quantum mechanics is the study of small matter, such as electrons, to understand it. 4 There Are Different Types of Electron Orbitals in Covalent Bondsįrontier Orbital Theory of Quantum Mechanics with Wave Functions.3.1 Conjugate Structure Changes the Energy and Spectrum of the HOMO-LUMO Gap.2.1 π Bonds and π* Bonds When Making Double and Triple Bonds with p Bonds.2 HOMO and LUMO by σ and σ* Bonds in p Orbitals.1.3 The Reason Why Helium Doesn’t Become a Molecule Has to Do with Its Anti-Bonding Orbital.1.2 The Anti-Bonding Orbitals (LUMO) Have Nodes and High Energy.1.1 Difference Between Bonding and Anti-bonding Orbitals.1 Frontier Orbital Theory of Quantum Mechanics with Wave Functions.Free rotation of atoms around pi bonds is not possible because it involves breaking the pi bonds. There can be free rotation of atoms around the sigma bonds. In other words, a single bond cannot be a pi bond. Thus, a pi bond is always present in molecules with multiple bonds, i.e., double or triple bonds. 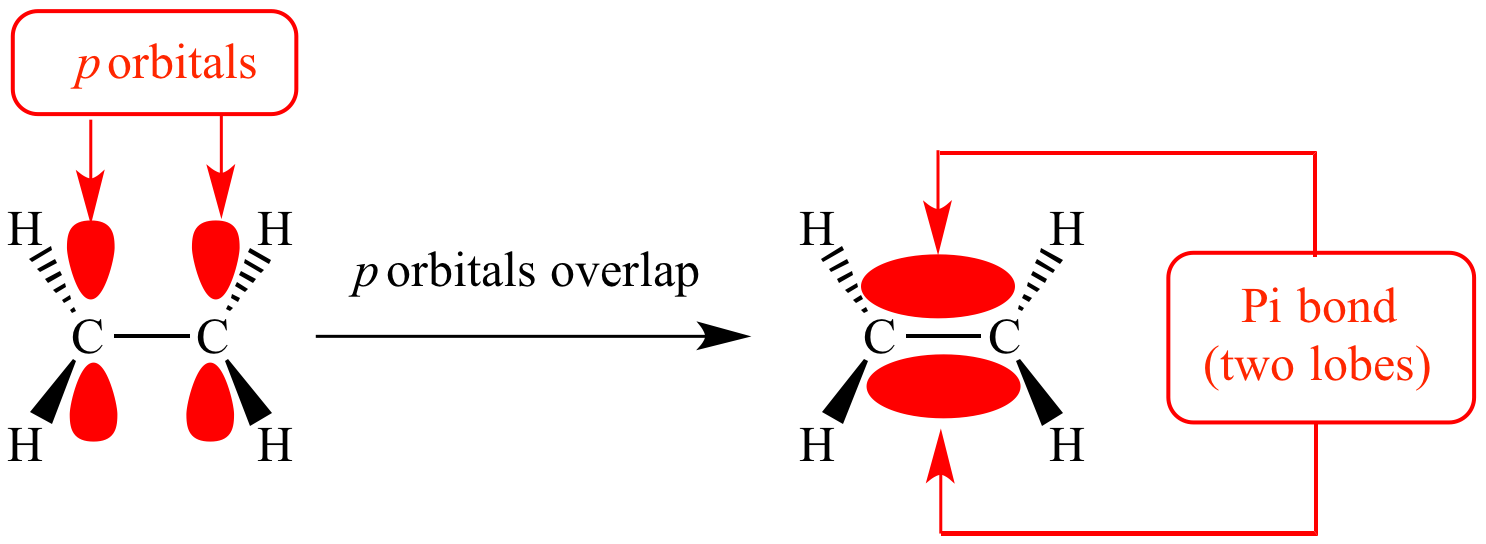
The reason is that the atoms constituting a single bond prefer to form a strong sigma bond rather than a weak pi bond. The reason is that the overlapping of atomic orbitals can take place to a greater extent during the formation of a sigma bond, whereas overlapping of orbitals occurs to a smaller extent during the formation of a pi bond.Ī pi bond between two atoms is formed only in addition to a sigma bond. The orbital overlap takes place in such a way that their axes are parallel to each other but perpendicular to the internuclear axis.Ī sigma bond is stronger than a pi bond. This type of covalent bond is formed by the lateral or sideways overlap of the atomic orbitals. 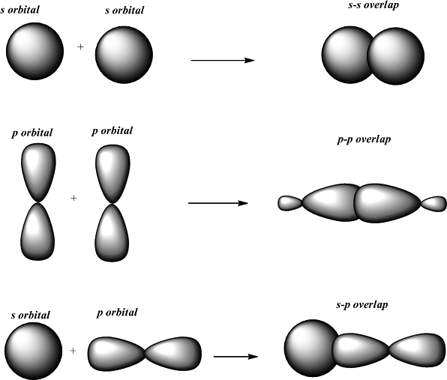
The atomic orbitals overlap along the inter-nuclear axis and involve end-to-end or head-on overlap. This type of covalent bond is formed by the axial overlapping of half-filled atomic orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
